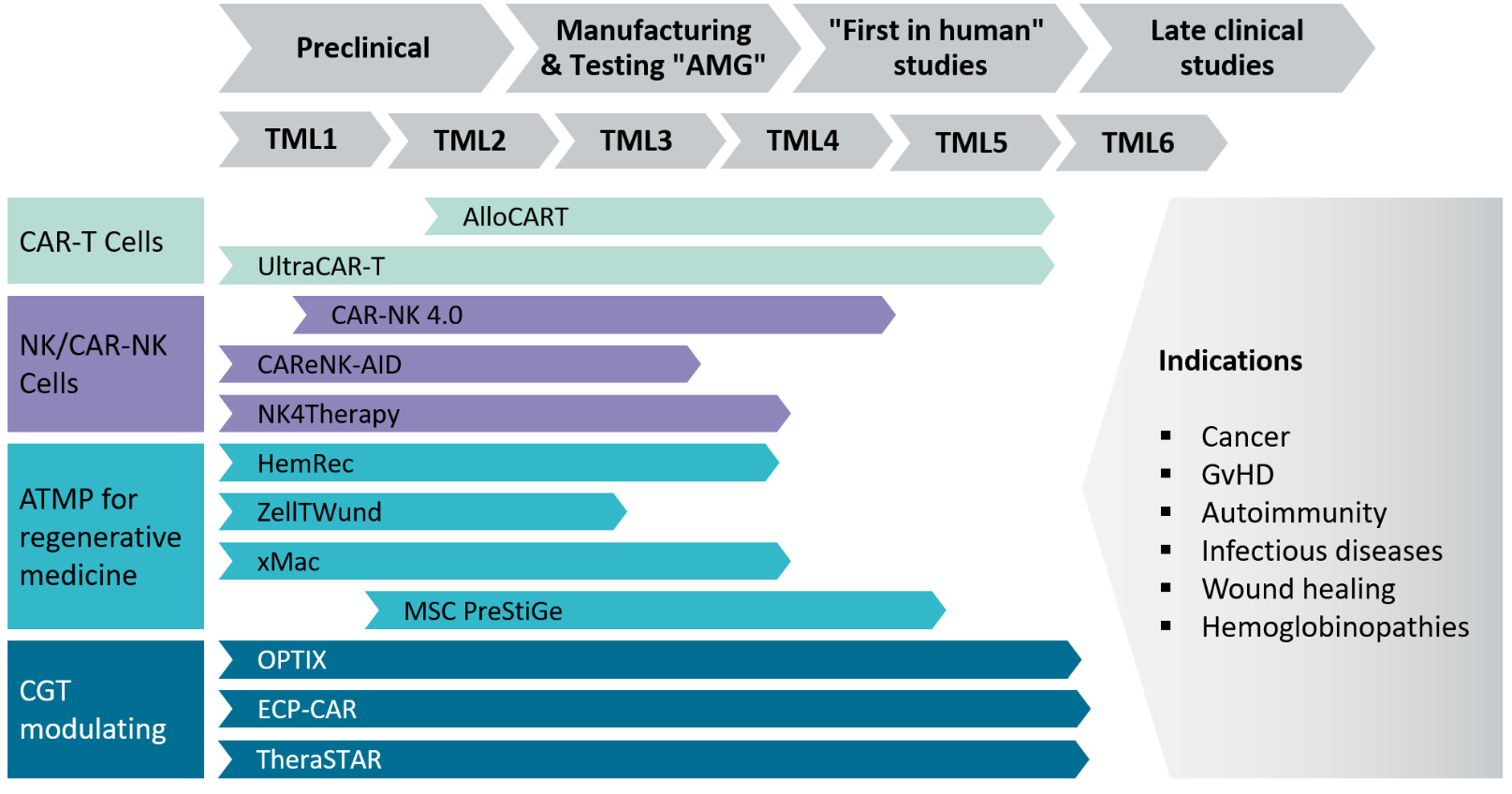
Selected research projects
Based on internal and external reviews, twelve innovative projects were selected from many submissions in a competitive process and divided into four areas:
Area 1 – CAR-T Cells: AlloCARTreg and UltraCAR-T
Area 2 – NK- and CAR-NK Cells: CAR-NK 4.0, CAReNK-AID and NK4Therapy
Area 3 – ATMPs for regenerative medicine: HemRec, ZellTWund, xMac and MSC-PreStiGe
Area 4 – CGT modulating: OPTIX, ECP-CAR and TheraSTARClinical application areas addressed by the projects include tumor diseases, graft-versus-host diseases (GvHD), autoimmune diseases, chronic wounds and congenital genetic diseases such as hemoglobinopathies.
Area 1: CAR-T Cells
AlloCARTreg
AlloCARTreg is aiming at the development of an universal off-the-shelf immunosuppressive cell product for treatment of autoimmune and inflammatory diseases. The project bundles cutting-edge adaptor CAR technology invented in the Bachmann/Feldmann lab, expertise from automated clinical grade manufacturing of T regulatory cells (Fuchs) and their clinical application (Bornhäuser), and state-of-the-art tools for (epi-) genome modifications (Buchholz) to enhance target specificity and facilitate allogeneic application.
Project lead: Prof. Dr. Martin Bornhäuser
UltraCAR-T
The project UltraCAR-T is dedicated to the core topic of cellular immunotherapy with genetically modified CAR-T cells using Sleeping Beauty technology based on a proof of concept between University Würzburg, Max Delbrück Center for Molecular Medicine and Fraunhofer IZI. ROR1 represents a tumor antigen validated by the project leader himself, which should enable a broad application in different tumor entities. Within the SaxoCell® Future Cluster, the value chain for ROR1 CAR-T cells is being developed and exploited in cooperation with the start-up T-CURX GmbH.
Project lead: Prof. Dr. med. Michael Hudecek
Area 2: NK- and CAR-NK Cells
CAR-NK 4.0
The project CAR-NK 4.0 focuses on the development of an AI-assisted CAR-NK cell platform (CAR-4.0) and an autonomous, fully automated manufacturing process with the goal of enabling a phase I trial of an allogeneic CD123-addressing CAR-NK approach for the treatment of myeloid neoplasms. To increase the therapeutic efficiency of this CAR-NK cell therapy and its manufacturing, novel technologies such as a bi-specific target approach, AAV gene transfer technologies and a new cell activation approach will be implemented.
Project lead: Prof. Dr. Dr. Ulrike Köhl
CAReNK-AID
The project CAReNK-AID focuses on the development of CAR-NK cell preparations targeting B lymphocytes and their precursors for novel applications such as severe autoimmune diseases. The project involves local intellectual property (IP) and experience with NK cells, novel gene transfer systems and automated processes.
Project lead: Prof. Dr. med. Torsten Tonn
NK4Therapy
The project NK4Therapy exploits the potential of a subset of memory NK cells for the immunotherapy of tumors and could also be used to treat the reactivation of viruses after an immunocompromising therapy, such as stem cell transplantation or acute viral infections. A technology patented by the TU Dresden for the production of this NK cell population will be used and brought to market maturity with a Saxon SME partner.
Project lead: Prof. Dr. Achim Temme
Area 3: ATMPs for regenerative medicine
HemRec
The project HemRec is based on the development of proprietary designer recombinases by the group of Prof. Dr. Buchholz (TUD). The platform technology itself is content of the newly founded TUD spin-off RecTech. In the present project, this lighthouse technology of SaxoCell® will be used for corrective regenerative therapy in hemoglobinopathies together with DKMS Life Science Lab GmbH. The strength of the technology lies in the safe and flexible genome editing compared to other related methods (e.g. CRISPR/Cas).
Project lead: Prof. Dr. Frank Buchholz
ZellTWund
The project ZellTWund is based on the generation and clinical use of purified pro-regenerative cells (in particular fibroblast subpopulations) from human skin as an approach for skin regeneration in chronic, non-healing wounds. The therapeutic approach uses degradable biopolymers that integrate pro-regenerative autologous stromal cells. The project has already generated interest from an industrial partner (NextCell Pharma, Sweden) and the partners intend a joint development within the SaxoCell® cluster.
Project lead: Prof. Dr. med. Jan Christoph Simon
xMac
The project xMac is based on the scientific discoveries of Prof. Sieweke’s group on the expansion potential of adult macrophages, as well as the differentiation and expansion of macrophages from human induced pluripotent stem cells. Macrophages have great potential as a broadly applicable future cell therapeutics. During the first project phase, the focus is on the development of a manufacturing process under GMP conditions to provide investigational products for clinical trials in the field of infectious and pulmonary diseases, as well as on the development of universally applicable preparations for allogeneic transplantation.
Project lead: Prof. Dr. Michael H. Sieweke
MSC-PreStiGe
The project MSC-PreStiGe exploits the immunomodulatory potential of mesenchymal stromal cells (MSC) extracted from umbilical cord tissue. This juvenile MSC population is particularly characterized by an immense expansion potential, which allows the treatment of up to 25 patients with only one cell product. A first indication to be tested for the use of this preparation is severe acute graft-versus-host disease (GvHD) after allogeneic stem cell transplantation.
Project lead: Prof. Dr. Mario Rüdiger
Area 4: Modulating ZGT
OPTIX
The project OPTIX addresses the optimization and clinical translation of a manufacturing process for an antibody-modified stem cell transplant (Palintra®) followed by a prospective, multicenter, single-arm first-in-man phase I/II trial in cooperation with the Saxon company Tcell Tolerance GmbH, as well as translational research to further elucidate the mechanism of action. This comprises a modification of an allogeneic hematopoietic stem cell transplant for the treatment of hematologic neoplasms, whereby the modification is intended to reduce graft-versus-host disease while preserving the graft-vs-leukemia effect.
Project lead: Lilly Stahl
ECP-CAR
In the project ECP-CAR, the additive effect of immunomodulation by extracorporeal photophoresis (ECP) will be tested in a phase II trial. Relevant endpoints will be the safety and efficacy of clinically established CAR-T cell therapeutics.
Project lead: Dr. med. Vladan Vucinic
TheraSTAR
The project TheraSTAR aims to develop theranostic targets (targets for therapy and diagnostics) that significantly support both conventional CAR and universal adapter CAR (UniCAR) technologies in the SaxoCell® cluster. In this context, a targeted molecule design is expected to enable cell-therapeutically beneficial modulation of the tumor milieu. In addition, the theranostic approach allows the monitoring of the progress of the immunotherapy in patients using imaging techniques (PET/SPECT) as well as endogenous radionuclide therapy.
Project lead: Dr. Anja Feldmann